1. Current Research and Principal Research Interests
We are interested in development of a new class of N-heterocyclic (NHC) ligands with saccharides concerning with water soluble catalysts and syntheses of new drugs. We are also studying reactions, especially oxidation, of small molecules as a ligand in organometallic compounds.
(1) Saccharide incorporated NHC ligands.
"Glucose incorporated NHC complexes":
An acetylglucose incorporated NHC precursor was synthesized by the reaction of acetobromoglucose and 1-methylimidazole. The precursor was reacted with Ag2O affording acetylglucose incorporated NHC Ag complexes, from which the NHC ligand was transferred to an Ir complex.
| 
|
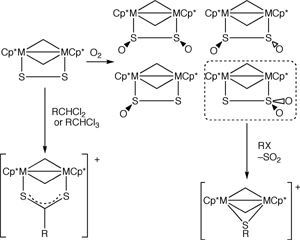
(2) Reactions of sulfide ligands bound on a rhodium or iridium dimetallic unit.
"Oxygenation of disulfide":
A disulfide ligand bound on the rhodium or iridium dimetallic unit
exhibits high reactivity towards molecular oxygen and resulted in the
formation of SSO, cis-OSSO, trans-OSSO, and
SSO2 complexes. Further oxygenation were introduced by
m-chloroperbenzoic acid to afford an
S(O)2SO2 complex. These results provide
information for understanding the sulfur cycle in nature.
"C-S bond formation on the dimetallic unit": The
SSO2 dirhodium or diiridium complexes react with
hydrocarbyl halides resulted in C-S bond formation accompanied with
genaration of SO2 gas. The S2 complexes also
reacted with hydrocarbylhalides to afford
hydrocarbyldithiocarboxylato complexes via C-H and C-Cl activation.
| 
|
(3) Oxygenation of rhodium complexes with cyclictridentate polypyridine ligand.
Rhodium mononuclear complexes with thiacalix[3]pyridine
([Rh(Py3S3)(diene)]+) are one of the
examples for rare cyclic polypyridine complexes. These
Py3S3 complexes are amphiphilic and used for
the investigation of their oxidation reaction both in water and an
organic solvent.
"Oxygenation by m-CPBA":
Sulfur bridges of the Py3S3 ligand in these
complexes were selectively oxygenated by m-CPBA in
dichloromethane to give sulfinylcalix[3]pyridine complexes,
[Rh{Py3(SO)3}(diene)]+, in which all three
oxygen atoms of the SO groups occupy the equatorial positions. The
oxygenation of the sulfur atoms in the ligand proceeded stepwise and
further oxygenation of the SO moiety occurred only for the nbd
complex having the smaller diene ligand resulting
[Rh{Py3(SO)2(SO2)}(nbd)]+.
"Oxygenation by H2O2":
The oxidation of [Rh(Py3S3)(cod)]+ by
H2O2 in water did not result oxygenation of the
sulfur bridges but the cod ligand hydoroxygenated to give
1,4,5,6-η4-2-hydroxycycloocta-4-ene-1,6-di-yl. This
is the result of oxygen activation by the rhodium ion.
| 
|
2. Selected Publications
1. "Sugar Incorporated N-Heterocyclic Carbene Complexes.", T. Nishioka, T. Shibata, I. Kinoshita,
Organometallics, 26, 1126-1128 (2007).
2. "Observation of Halide Induced Conformational Conversion of Dinuclear Cu Complexes Having a Tetradentate Polypyridine Ligand with a p-Xylene Backbone.", T. Nishioka, S. Mitsui, I. Kinoshita, T. Koshiyama, M. Kato,
Bull. Chem. Soc. Jpn., 80, 1357-1367 (2007).
3. "Thiacalix[3]pyridine produces a stable mononuclear rhodium(II) complex with mutual Jahn-Teller effect.", R. H. Tanaka, T.
Nishioka, I. Kinoshita, T. Takui, R. Santo, A. Ichimura,
Dalton Trans., 2006, 1374-1376.
4. "Direct Observation of Photochromic Dynamics in Crystalline-state of an Organorhodium Dithionite Complex.", H. Nakai, M. Mizuno, T. Nishioka, N. Koga, K. Shiomi, Y. Miyano, M. Irie, B. K. Breedlove, I. Kinoshita, Y. Hayashi, Y. Ozawa, T. Yonezawa, K. Toriumi, K. Isobe,
Angew. Chem., Int. Ed. Eng., 45, 6473-6476 (2006).
5. "Diamagnetic-Paramagnetic Conversion of Tris(2-pyridylthio)methylcopper(III) through a Structural Change From Trigonal Bipyramidal to Octahedral.", R. Santo, R. Miyamoto, R. Tanaka, T. Nishioka, K. Sato, K. Toyota, M. Obata, S. Yano, I. Kinoshita, A. Ichimura, T. Takui,
Angew. Chem., Int. Ed. Eng., 45, 7611-7614 (2006).
6. "Selective oxygenation of amphiphilic thiacalix[3]pyridine Rh(I)
diene complexes in both water and organic solvents", T.
Nishioka, Y. Onishi, K. Nakajo, G.-X. Jin, R. Tanaka, I. Kinoshita,
Dalton Trans., 2005, 2130-2137.
7. "A complete series of copper(II) halide complexes (X = F, Cl, Br,
I) with a novel CuII-C(sp3) bond", R. Miyamoto,
R. Santo, T. Matsushita, T. Nishioka, A. Ichimura, Y. Teki, I.
Kinoshita,
Dalton Trans., 2005, 3179-3186.
8. "A two-dimensional clathrate hydrate sandwiched by planar arrays
of a copper complex", R. Miyamoto, R. T. Hamazawa, M. Hirotsu, T.
Nishioka, I. Kinoshita, L. J. Wright,
Chem. Commun.,
2005, 4047-4049.
9. "Catalytically Active μ-Oxodiiron(IV) Oxidants from Iron(III)
and Dioxygen", A. Ghosh, F. T. de Oliveria, T. Yano, T. Nishioka, E. S. Beach, I. Kinoshita, E. Münck, A. D. Ryabov, C. P. Horwitz, T. J. Collins,
J. Am. Chem. Soc., 127,
2505-2513 (2005).
10. "Nucleophilicity of Ligated S22- Ions.
Conversion of Organic Chlorides into Organosulfur Compounds in
cis-[(MCp*)2(μ-CH2)2(μ-S2R)]+ (M = Rh, Ir)", T. S. Lobana, K. Isobe, H. Kitayama, T. Nishioka, M. Doe, I. Kinoshita.
Organometallics, 23, 5347-5352 (2004).
11. "Novel Nucleophilic Reactivity of Disulfide Ligands Coordinated
Parallel to M-M (M = Rh, Ir) Bonds", T. Nishioka, H. Kitayama,
B. K. Breedlove, K. Shiomi, I. Kinoshita, K. Isobe,
Inorg. Chem., 43, 5688-5697 (2004).
12. "Activation of C-Cl and C-H bonds by Ligated S22- in Unusual Way. Conversion of Organic Chlorides into Organosulfur Compounds Coordinated to Iridium Metal Atom in cis-[(IrCp*)2(μ-CH2)2(μ-S2R)]+", T. S. Lobana, K. Isobe, H. Kitayama, T. Nishioka, I. Kinoshita,
Angew. Chem., Int. Ed. Eng., 43, 213-215 (2004).
13. "Design and synthesis of copper complexes of novel ligands based
on the pyridine thiolate group", I. Kinoshita, L. J. Wright, S. Kubo,
K. Kimura, A. Sakata, T. Yano, R. Miyamoto, T. Nishioka, K. Isobe.
Dalton Trans., 2003, 1993-2003.
14. "Structure and reactivity of a novel parallel thiosulfito (SSO2-S:S') rhodium dinuclear complex ",K. Shiomi, B. K. Breedlove, H. Kitayama, T. Nishioka, I. Kinoshita, N. Koga, K. Isobe,
Chem. Commun., 2002, 1756-1757.
15. "Thia-calix[n]pyridines, synthesis and coordination to Cu(I,II) ions with both N and S donor atoms", R. Tanaka, T. Yano, T. Nishioka, K. Nakajo, B. K. Breedlove, K. Kimura, I. Kinoshita, K. Isobe,
Chem. Commun., 2002, 1686-1687.
|



